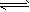已知:H2(g)+12O2(g)=H2O(g)△H=-242kJ/mol,断开1molO=O键和1molH-O键所吸收的能量分别为496kJ和463kJ,则断开1molH-H键所吸收的能量为()A.9
2023-04-26
问题描述:
已知:H2(g)+| 1 |
| 2 |
A.920 kJ
B.557 kJ
C.436 kJ
D.188 kJ
最佳答案:
1 molH2O中含2molH-O键,断开1 mol O=O键和1 molH-O键所吸收的能量分别为496 kJ和463kJ,令H-H键的键能为x,对于反应H2(g)+12O2(g)=H2O(g)△H=-242kJ/mol,反应热=反应物的总键能-生成物的总键能,故△H=xkJ/m...






热门标签